 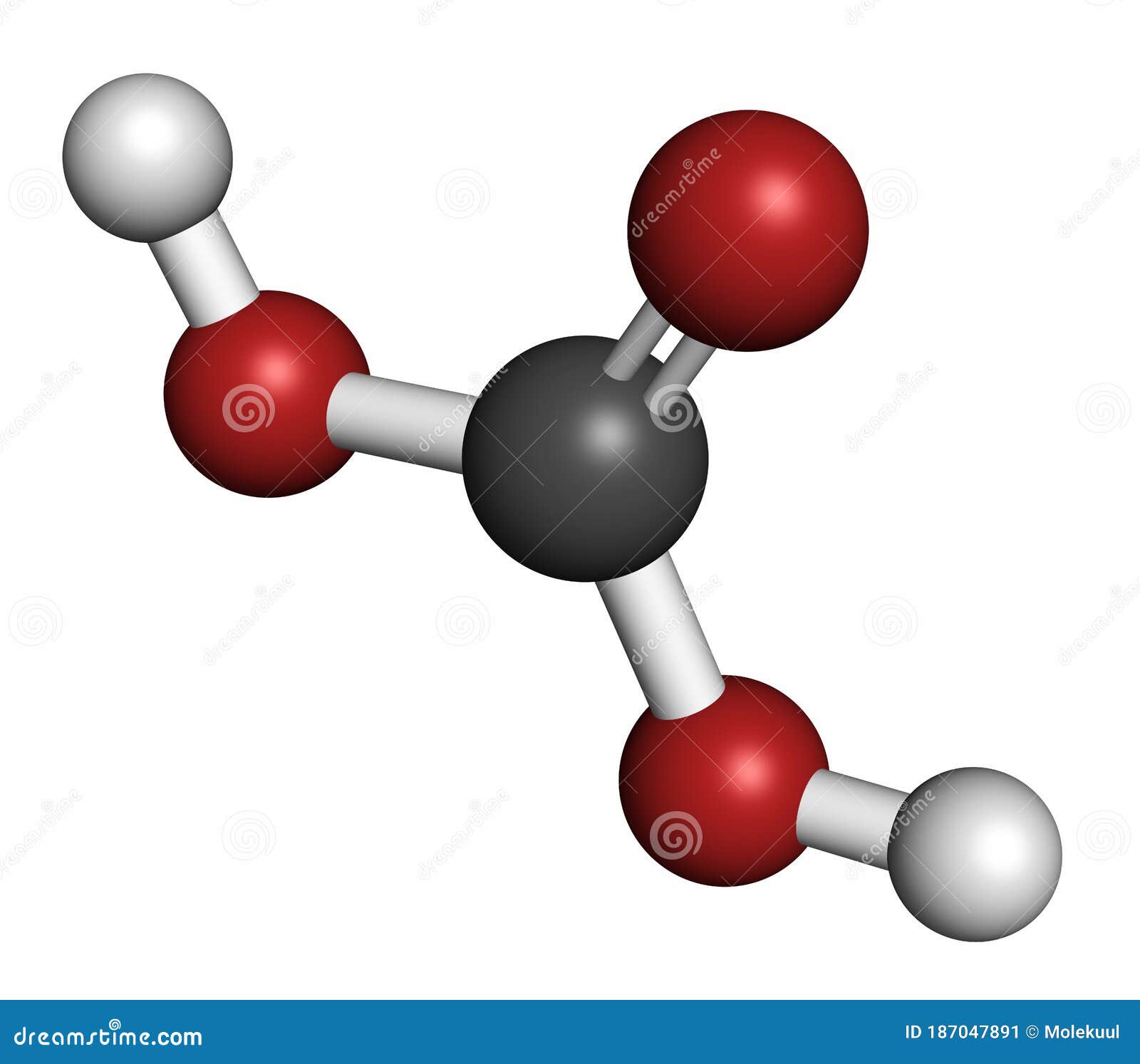 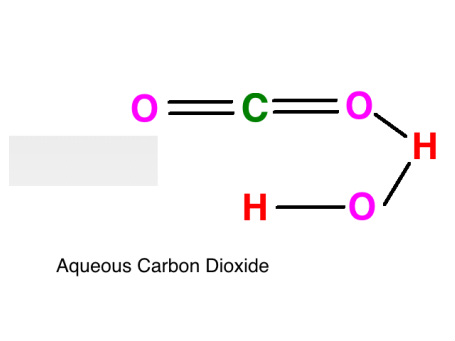
Raven JA, Giordano M, Beardall J, Maberly SC (2012) Algal evolution in relation to atmospheric CO 2: carboxylases, carbon-concentrating mechanisms and carbon oxidation cycles. Moyle JB (1946) Some indices of lake productivity. Mandal BK, Boyd CE (1980) The reduction of pH in water of high total alkalinity and low total hardness. Professional paper 440-G, United States Government Printing Office, Washington, DC Livingstone DA (1963) Chemical composition of rivers and lakes. Korb RE, Saville PJ, Johnston AM, Raven JA (1997) Sources of inorganic carbon for photosynthesis by three species of marine diatom. Water-supply paper 1473, United States Geological Survey, United States Government Printing Office, Washington, DC Hem JD (1970) Study and interpretation of the chemical characteristics of natural water. Freeman, Cooper, and Company, San Francisco Garrels RM, Christ CL (1965) Solutions, minerals, and equilibria. US Bur Fish Bull 22:367–437įrear CL, Johnston J (1929) The solubility of calcium carbonate (calcite) in certain aqueous solutions at 25☌. American Public Health Association, Washington, DCĮllis MM (1937) Detection and measurement of stream pollution. Aquat Bot 98:57–60Įaton AD, Clesceri LS, Rice EW, Greenburg AE (eds) (2005) Standard methods for the examination of water and wastewater. N Am J Aquac 73:403–408Ĭavalli G, Riis T, Baattrup-Pedersen A (2012) Bicarbonate use in three aquatic plants. Bulletin 465, Alabama Agricultural Experiment Station, Auburn University, Auburnīoyd CE, Tucker CS, Viriyatum R (2011) Interpretation of pH, acidity and alkalinity in aquaculture and fisheries. Craftmaster Printers, Auburnīoyd CE, Walley WW (1975) Total alkalinity and hardness of surface waters in Alabama and Mississippi. Proc Annu Conf Southeast Assoc Game Fish Comm 20:413–424īoyd CE, Tucker CS (2014) Handbook for aquaculture water quality. Geochim Cosmochim Acta 29:343–352īoyd CE, Lawrence JM (1966) The mineral composition of several freshwater algae. The solubility of calcite in relation to ionic strength. KeywordsĪkin GW, Lagerwerff JV (1965) Calcium carbonate equilibria in aqueous solution open to the air. Fish and other aquatic animals avoid high carbon dioxide concentration, but 20 mg/L or more can be tolerated if there is plenty of dissolved oxygen. The optimum pH range for most aquatic organisms is 6.5–8.5, and the acid and alkaline death points are around pH 4 and pH 11, respectively. Water bodies with moderate to high alkalinity are well-buffered against wide daily swings in pH resulting from net removal of carbon dioxide by photosynthesis during daytime and return of carbon dioxide to the water by respiratory process at night when there is no photosynthesis. Alkalinity increases the availability of inorganic carbon for photosynthesis, because most phytoplankton species and many higher aquatic plants can obtain carbon from bicarbonate. Total alkalinity typically is less than 50 mg/L in waters of humid areas with highly leached soils, but it is greater where soils are more fertile, limestone formations are present, or the climate is arid. The total concentration of titratable bases-usually bicarbonate and carbonate-expressed in milligrams per liter of calcium carbonate is the total alkalinity. Limestone, calcium silicate, and feldspars in soils and other geological formations dissolve through the action of carbon dioxide to increase the concentration of bicarbonate in water and raise the pH. 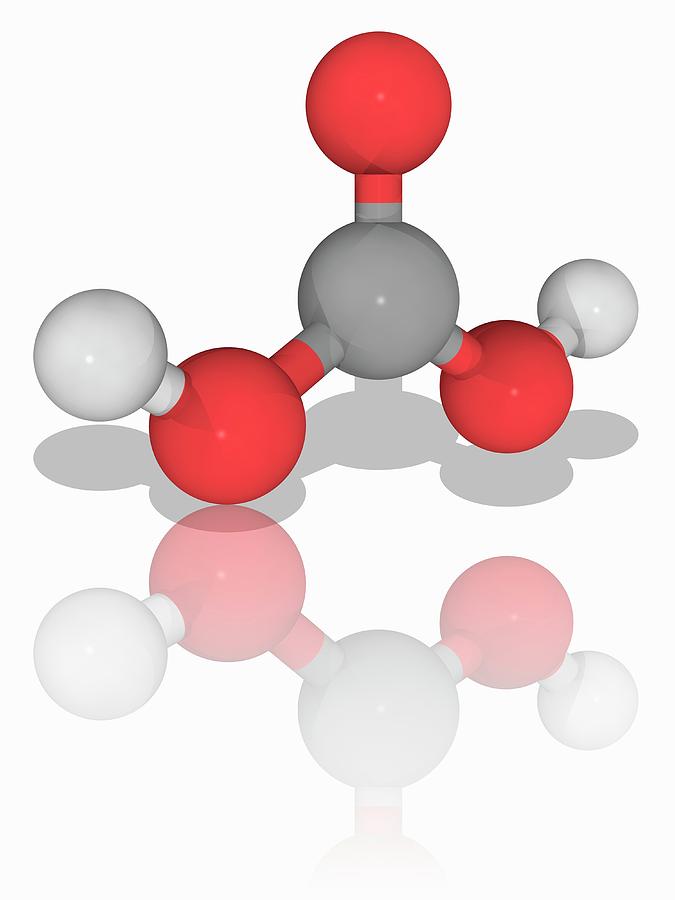
Because dissolved carbon dioxide is acidic, rainwater that is saturated with this gas is naturally acidic-usually about pH 5.6. The pH or negative logarithm of the hydrogen ion concentration is a master variable in water quality because the hydrogen ion influences many reactions. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
